FRDD
Forensically-Relevant Drug Database

Description
Preferred Drug Name
Brief Description
The following information was compiled in June 2023 and is subject to change as new research is conducted and as new information becomes available: Description: N-Pyrrolidino protonitazene is a novel synthetic opioid bearing structural resemblance to protonitazene, N-pyrrolidino etonitazene, and other nitazene (2-benzylbenzimidazole) analogues and is an isomer of N-pyrrolidino isotonitazene. In January 2023, N-pyrrolidino protonitazene was detected for the first time in the United States. Our laboratory continues to gather toxicology data for this novel drug. Sample Source: NMS Labs – Toxicology Laboratory, NMS Labs – Criminalistics Laboratory, etc. Sample Appearance: Drug Material – Brown Powder. Toxicology – Blood Specimens. Pharmacology: Limited information regarding the pharmacology of N-pyrrolidino protonitazene is currently known. Recent in vitro studies examining activity and potency found that N-pyrrolidino protonitazene is an active opioid with potency approximately 25 times greater than that of fentanyl [unpublished data from L. De Vrieze and C. Stove]. Based on structural similarity, N-pyrrolidino protonitazene is expected to exhibit similar adverse effects to other nitazene analogues. Toxicology: N-Pyrrolidino protonitazene has been detected in eight toxicology cases at the CFSRE. Drug Materials: N-Pyrrolidino protonitazene has been identified in one drug material at the CFSRE. Demographics / Geographics: Cases originated from at least three states, including Illinois, West Virginia, and Minnesota. In death investigations, decedents were predominantly male, ranging in age from 20s-70s. Legal Status: N-Pyrrolidino protonitazene is not explicitly scheduled in the United States.
Structure
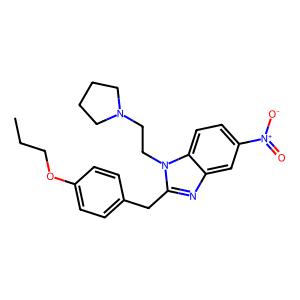
Nomenclature
Formal Name (IUPAC)
Synonyms
Identifiers
CAS Numbers
Chemistry
Chemical Formula
Molecular Mass
Molecular Ion
Exact Mass
SMILES
InChI String
InChI Key
GCMS Analytics
Lab
Comments
| Sample Preparation: | Sample: Acid / base extraction Standard (above): Diluted in methanol |
| Instrument: | Agilent 5975 Series GC/MSD |
| Methods: | http://www.cfsre.org/nps-discovery/monographs |
Images
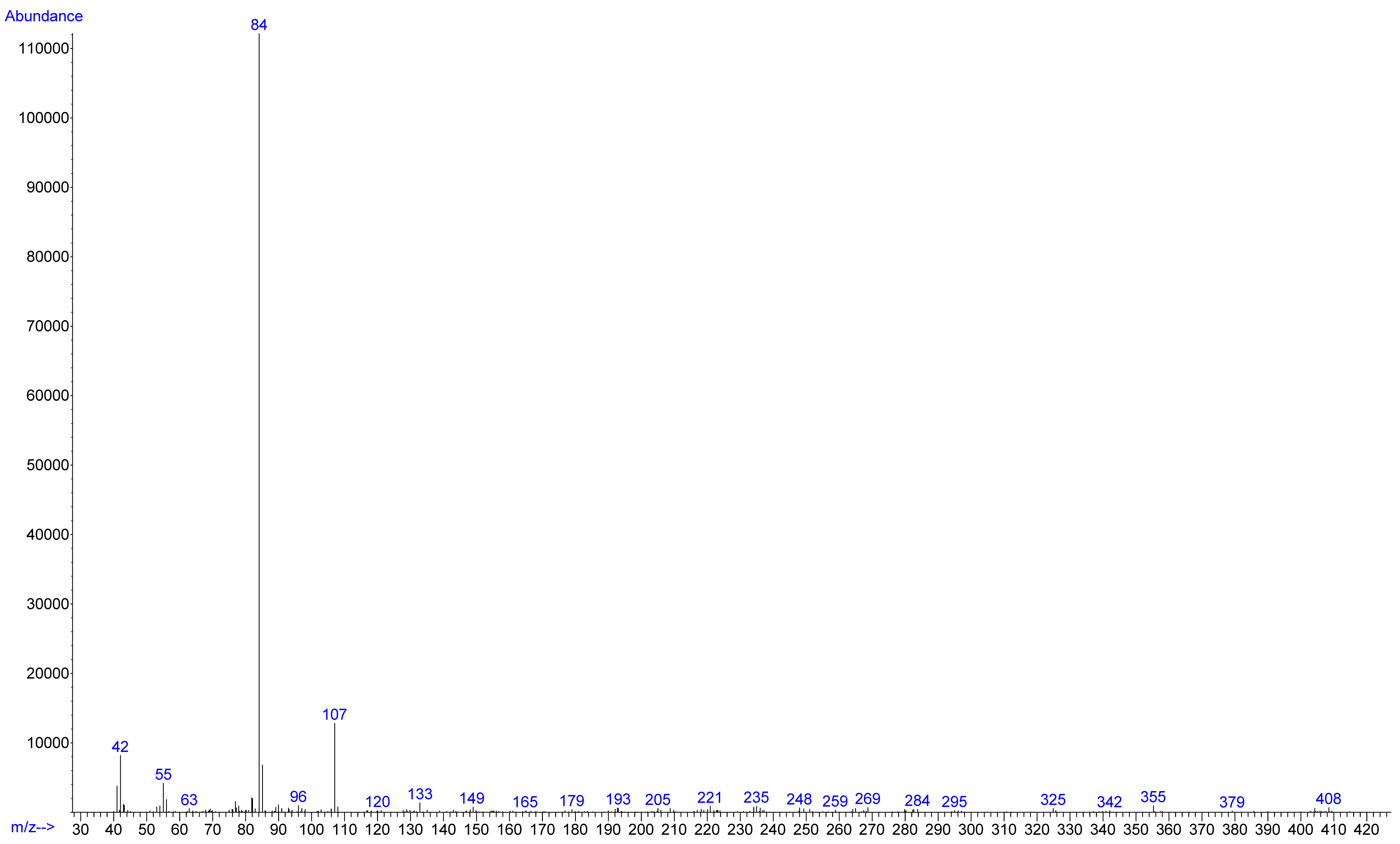
Fragments
107
42
HRMS Analytics
Lab
Comments
| Sample Preparation: | Acid / base extraction |
| Instrument: | Sciex TripleTOF® 5600+ LC-QTOF-MS |
| Methods: | https://www.cfsre.org/nps-discovery/monographs |
Images
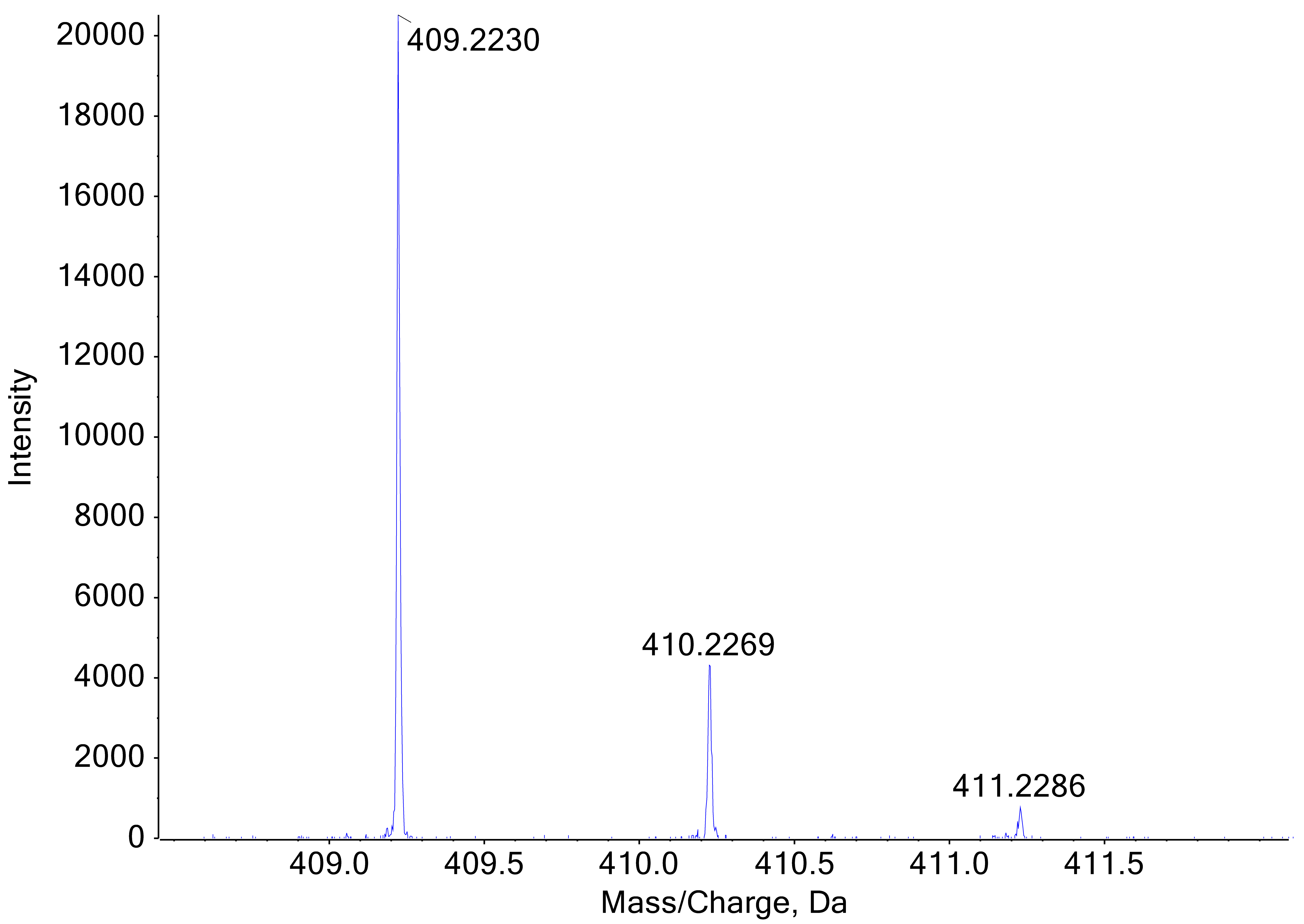
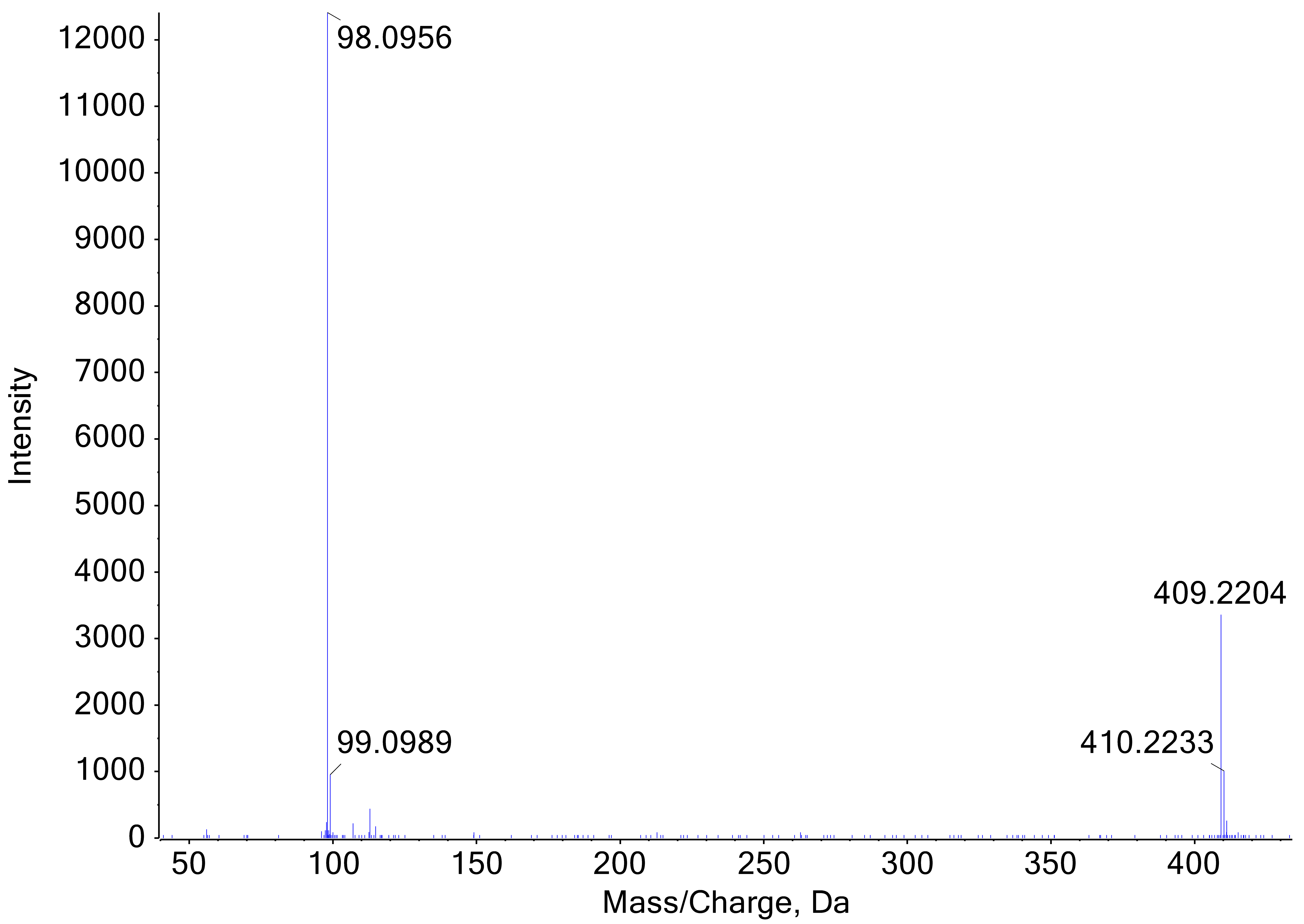
Fragments
409.223