FRDD
Forensically-Relevant Drug Database

Description
Preferred Drug Name
Brief Description
5,6-Dichloro brorphine is a novel synthetic opioid characterized as an “orphine” analogue and bears structural similarity to other benzimidazolones (e.g., brorphine, chlorphine, 5,6-dichloro desmethylchlorphine). 5,6-Dichloro brorphine was originally synthesized and researched for use for analgesia with a suggested large therapeutic window and decrease of severe adverse side effects (e.g., respiratory depression). 5,6-Dichloro brorphine was first identified by our laboratory in November 2025 and confirmed after acquiring standard reference material.
Structure
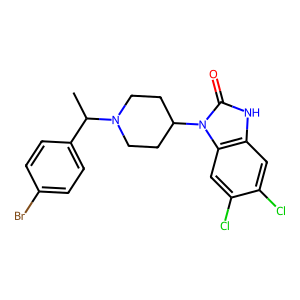
Nomenclature
Formal Name (IUPAC)
Synonyms
Identifiers
CAS Numbers
Chemistry
Chemical Formula
Molecular Mass
Molecular Ion
Exact Mass
SMILES
InChI String
InChI Key
GCMS Analytics
Lab
Comments
| Sample Preparation: | Drug Materials: Dilution in methanol or acid / base extraction Drug Standards: Dilution in methanol |
| Instrument: | Agilent 5975 Series GC/MSD System |
| Column: | Agilent J&W DB-1 (12 m x 200 µm x 0.33 µm) |
| Carrier Gas: | Helium (Flow: 1.46 mL/min) |
| Temperatures: | Injection Port: 265 °C Transfer Line: 300 °C MS Source: 230 °C MS Quad: 150 °C Oven Program: 50 °C for 0 min, 30 °C/min to 340 °C for 2.3 min |
| Injection Parameters: | Injection Type: Splitless, Injection Volume: 1 µL |
| MS Parameters: | Mass Scan Range: 40-550 m/z |
Images

Fragments
104
183
284
169
HRMS Analytics
Lab
Comments
| Sample Preparation: | Toxicology Samples: Liquid-liquid extraction Drug Materials: Dilution of sample in mobile phase |
| Instrument: | Sciex TripleTOF® 5600+, Shimadzu Nexera XR UHPLC Sciex X500R, Sciex ExionLC |
| Column: | Phenomenex® Kinetex C18 (50 mm x 3.0 mm, 2.6 µm) |
| Mobile Phase: | A: Ammonium formate (10 mM, pH 3.0) B: Methanol/acetonitrile (50:50) Flow rate: 0.4 mL/min |
| Gradient: | Initial: 95A:5B; 5A:95B over 13 min; 95A:5B at 15.5 min |
| Temperatures: | Autosampler: 15 °C Column Oven: 30 °C Source Heater: 600 °C |
| Injection Parameters: | Injection Volume: 10 µL |
| QTOF Parameters: | TOF MS Scan Range: 100-510 Da Precursor Isolation: SWATH® acquisition (27 windows) Fragmentation: Collison Energy Spread (35±15 eV) MS/MS Scan Range: 50-510 Da |
Images
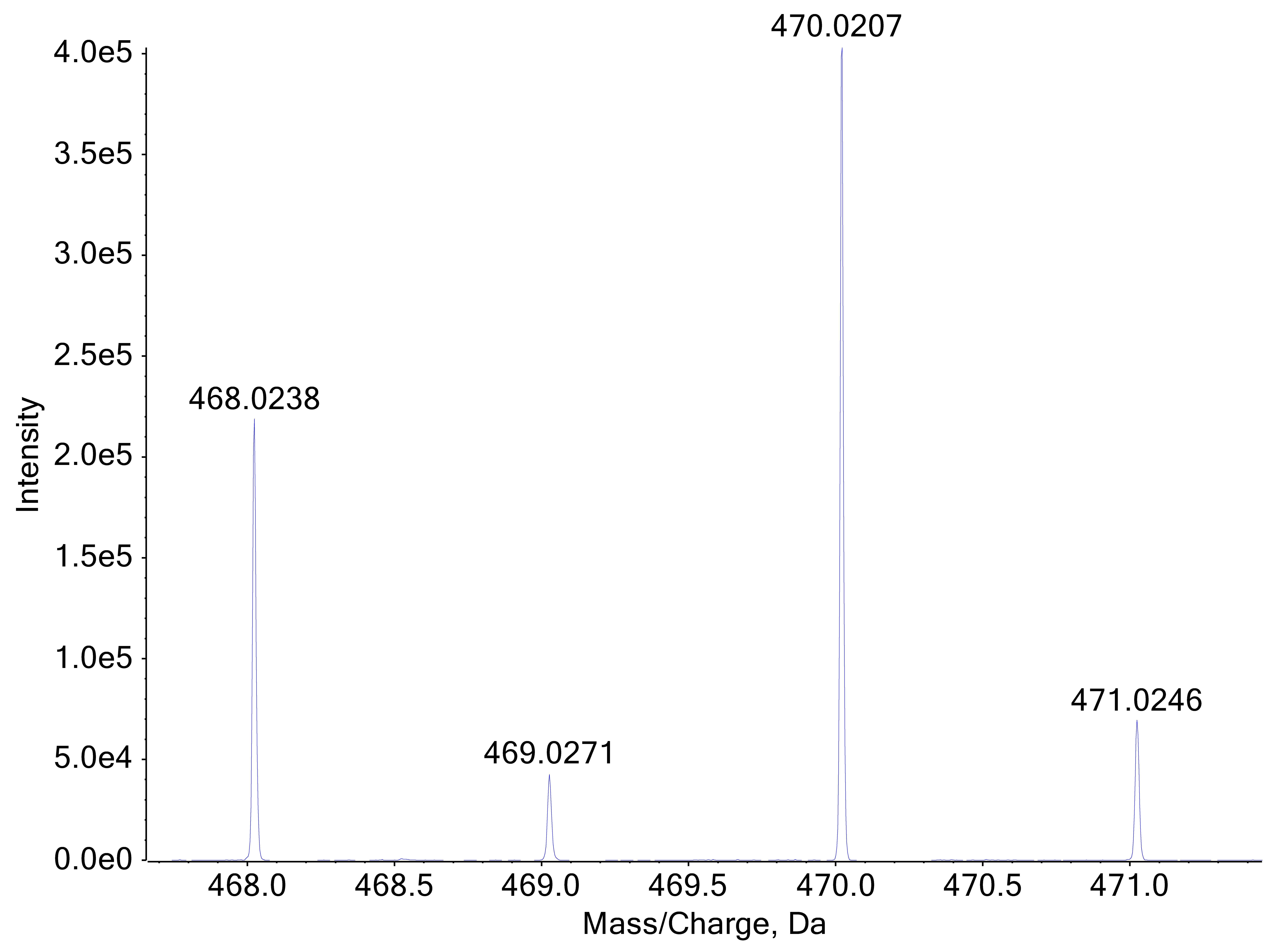
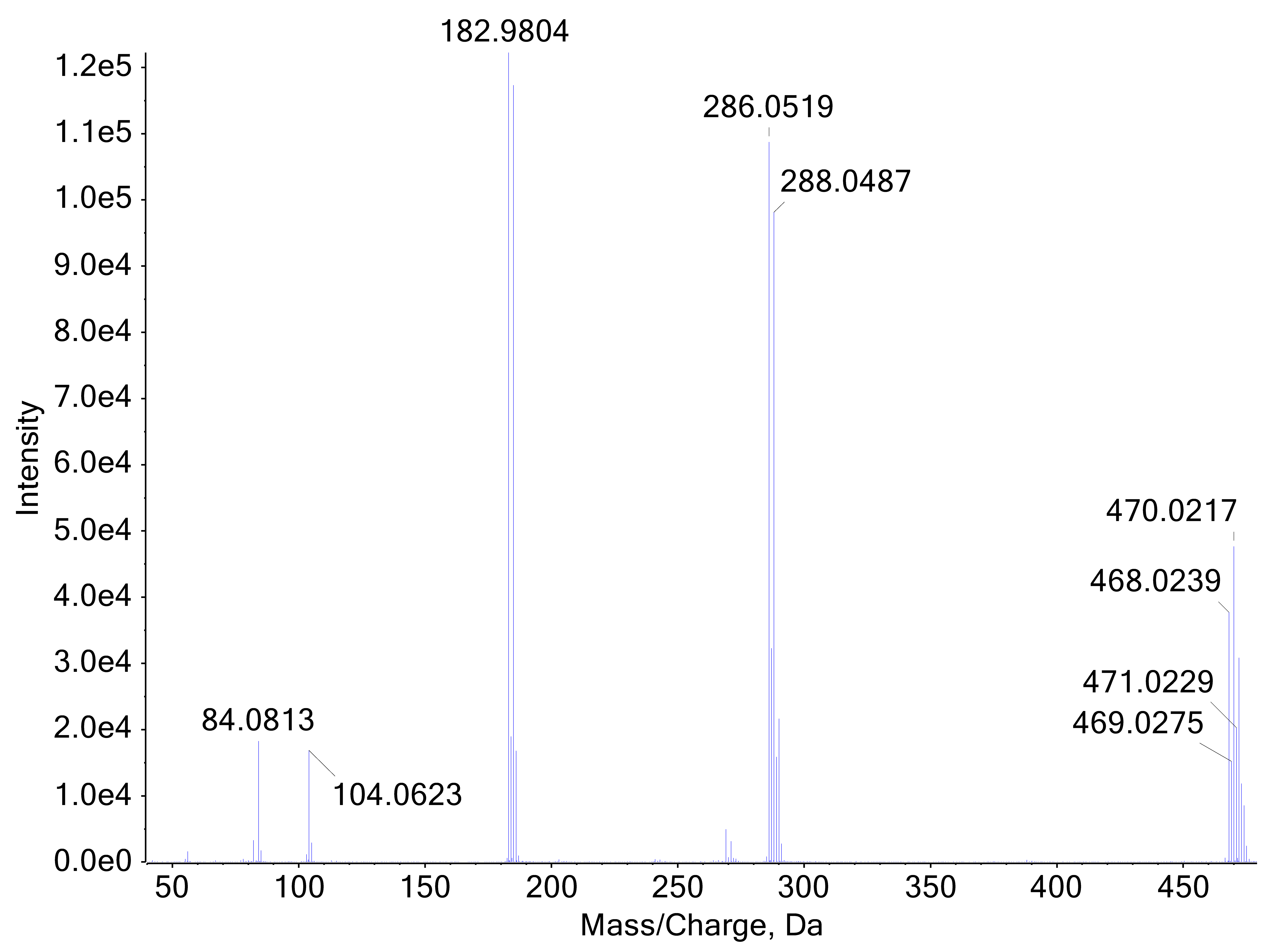
Fragments
286.0519
470.0217
84.0813
104.0623